

ALINAMIN A
アリナミンA
CHARACTERISTICS
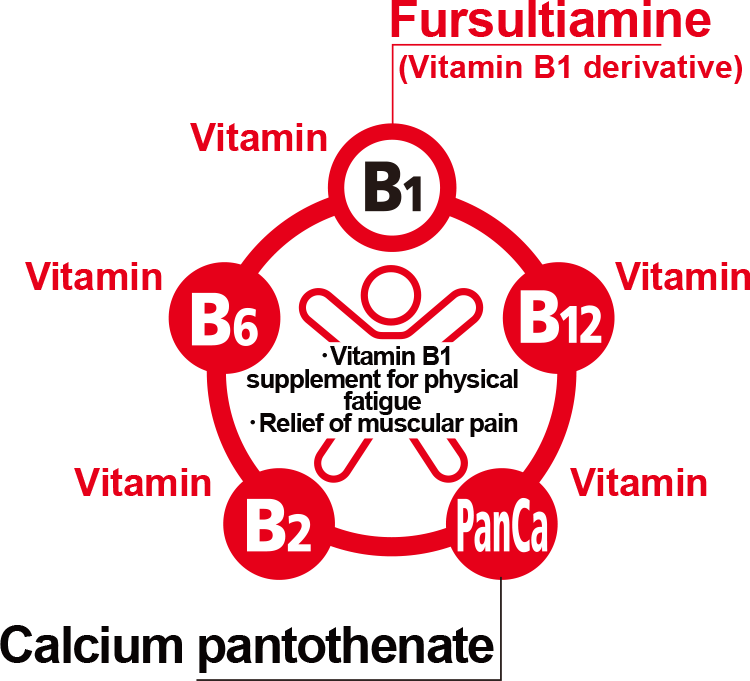
- ALINAMIN A contains fursultiamine, a highly absorbable vitamin B1 derivative, and is remarkably effective in relieving fatigue or alleviating the sense of feeling languid or heavy in everyday life.
- In addition to containing fursultiamine, vitamin B2 and vitamin B6, this medicine contains calcium pantothenate, which becomes coenzyme A and plays an important role in the production of energy. These ingredients are remarkably effective in relieving fatigue by helping to effectively change the three major nutrients (fat, protein, carbohydrates) to energy.
- This medicine is a yellow, sugarcoated tablet, which is easy to swallow.
INDICATIONS
- Supply of vitamin B1 in the following cases:
Physical fatigue, during pregnancy or lactation, and loss of strength during or after illness. - Relief of the following symptoms:
Muscular pain or joint pains (lower back pain, stiff shoulders, frozen shoulder, etc.), neuralgia, numbness of the extremities, constipation, and eye fatigue. - Beriberi
If the symptoms described in points 2 and 3 do not improve after using this medicine for one month, consult your physician or pharmacist.
DOSAGE AND DIRECTIONS
Take the following amount of medicine without chewing immediately after a meal, with cold or lukewarm water.
| Age | One dose | Daily dose |
| 15 years or over | 1–3 tablets | Once |
| 11–14 years | 1-2 tablets | |
| 7-10 years | 1 tablet | |
| Under 7 years | Do not take | |
Precautions regarding dosage and directions
- Children are permitted to take this medicine only under the direction and supervision of a parent or other responsible adult.
- Please follow the recommended dosage and directions.
INGREDIENTS
In three tablets (maximum daily dose for 15 years or over)
| Ingredients | Contents |
|---|---|
| Fursultiamine (vitamin B1 derivative) | 100mg |
| (in fursultiamine hydrochloride equivalent, | 109.16mg) |
| Pyridoxine hydrochloride (vitamin B6) | 20mg |
| Cyanocobalamin (vitamin B12) | 60μg |
| Riboflavin (vitamin B2) | 12mg |
| Calcium pantothenate | 15mg |
Inactive ingredients
Calcium lactate hydrate, partly pregelatinized starch, hypromellose, cellulose, lactose hydrate, hydroxypropylcellulose, magnesium stearate, corn starch, meso-erythritol, titanium dioxide, acacia, calcium carbonate, talc, sucrose
Precautions regarding ingredients
Although urine may turn yellow after taking this medicine, do not worry because this is caused by riboflavin in this medicine.
PRECAUTIONS FOR USE
Who should seek consultation
- Stop taking this medicine immediately and consult a physician, pharmacist or registered salesperson* if the following symptoms appear after taking this medicine because they might be side effects of the medicine. Take this leaflet with you.
Affected area Symptoms Skin Rash, redness, itching Digestive organs Nausea, vomiting, stomatitis - Stop taking this medicine and consult a physician, pharmacist or registered salesperson if the following symptoms appear after taking this medicine and continue or worsen. Take this leaflet with you.
Soft stool, diarrhea - Stop taking this medicine and consult a physician, pharmacist or registered salesperson if symptoms do not improve after taking this medicine for about one month. Take this leaflet with you.
*Registered salesperson: person who has the license to sell OTC medicines except Category 1.
PRECAUTIONS FOR STORAGE AND HANDLING
Product in a bottle or unit dose packaging
- Keep out of the reach of children.
- Do not take this medicine after the expiration date.
Product in a bottle
- Keep the bottle tightly closed and put it in the box. Store the box in a dry, cool place away from direct sunlight.
- To prevent improper use and loss of quality, do not transfer the medicine to another container.
- Once the bottle is opened, throw away the filler in the bottle. (Putting the filler back into the bottle may change the quality by absorbing moisture. The filler in the bottle protects the tablets from breakage during transportation.)
- Be sure to close the cap tightly after each use. (Failure to tightly close the bottle may result in loss of the quality of the medicine due to moisture.)
- Fill in the date when the bottle was opened in the column provided on the box and bottle.
- Once the bottle is opened, take this medicine within about 6 months to maintain quality.
Product in unit dose packaging
- Store in a dry, cool place away from direct sunlight.
- When taken in divided doses, fold over the opened edge of the pouch and store it. The medicine should be taken within four days.
